Certificación
The Estrogen–Mitochondria Axis: Energy, Hormones, and Mental Health
Recent research is beginning to reframe mental health through the lens of cellular energy. Across mood disorders, mitochondrial function has emerged as a key factor in how the brain regulates stress, emotion, and resilience. Estrogen plays a central role in this process, supporting mitochondrial activity, reducing oxidative stress, and helping maintain neurobiological stability.
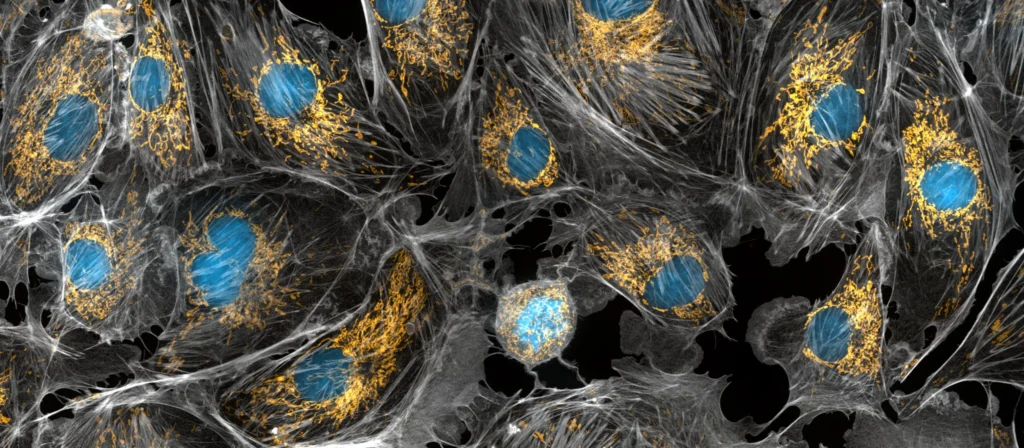
Image: Torsten Wittmann, UCSF (via NIH)
Mental health is influenced by a complex interplay of systems that do not operate in isolation, and is closely tied to how the body generates and uses energy. Mitochondria are central to this process; once viewed mainly as energy producers, they are now understood to help regulate immune signaling, stress responses, and brain function.
Mitochondrial function is sensitive to psychosocial influences such as chronic stress, trauma, and social connection, with downstream effects on inflammation and disease risk. A growing body of research links chronic stress with reduced mitochondrial efficiency, increased oxidative stress and inflammation, and disruptions in brain signaling.
Hormonal systems—particularly estrogen—further shape how these processes are regulated over time. Across the lifespan, transitions such as the menstrual cycle, postpartum period, and perimenopause bring shifts in estrogen signaling that extend beyond reproduction and can affect sleep, metabolism, mood, and cognition.
In the new edition of Nutrition Essentials for Mental Health, these connections are explored in greater depth, including the role of mitochondrial function and targeted nutritional strategies that support cellular energy and resilience.
The studies below examine how these interconnected systems contribute to patterns of vulnerability and adaptation in mental health.
The first study examines how fluctuations in estrogen may influence mitochondrial function in bipolar disorder. The authors describe bipolar disorder as a condition associated with dysregulated bioenergetics, immune changes, and metabolic dysfunction, and suggest that the estrogen–mitochondria axis may help explain sex-related differences in symptom patterns and periods of psychiatric vulnerability.
The second study explores how psychological experience connects to biology at the cellular level. It describes mitochondria as a key biological link between stress, immune activity, and brain function, showing how factors such as chronic stress, trauma, and social connection can influence mitochondrial processes, with downstream effects on inflammation and mental health, though some of this evidence is still emerging.
The third study examines how mitochondrial dysfunction contributes to mood disturbances during perimenopause. The authors describe these conditions as complex neuroendocrine and metabolic processes, where disruptions in mitochondrial homeostasis are associated with synaptic changes, neuroimmune activity, and increased inflammation. These shifts interact with fluctuating estrogen levels and are linked to symptoms such as depression, anxiety, and cognitive changes.
The fourth study expands this perspective by examining the relationship between estrogen, the gut microbiota, and mental health. The authors describe how fluctuations in estrogen can influence neurotransmitter balance, neuroinflammation, and mitochondrial function, while also shaping microbial diversity and metabolic activity. These changes may reinforce one another, increasing susceptibility to depressive symptoms through interactions along the gut–brain axis.
The fifth study takes a broader view of how mitochondria shape mental health. It links mitochondrial function to cognition, behavior, and a wide range of psychiatric conditions, while also emphasizing how sleep, nutrition, movement, and stress influence these processes over time.
Tags: mitochondria, mental health, estrogen, mitochondrial function, neurobiology, hormonal health, perimenopause, depression, inflammation, oxidative stress, gut brain axis, brain health, cellular energy, women’s health
Interested in Learning More?
- Course(s): Mental Health Disorders
- Course(s): NEW! Rhythms of Life: Aging and the Restoration of Psychobiological Coherence
- Book(s): Natural Woman: Herbal Remedies for Radiant Health at Every Age and Stage of Life
- Book(s): The Brainbow Blueprint: A Clinical Guide to Integrative Medicine and Nutrition for Mental Well-Being
Research Glossary
Research has its own vocabulary. To help you decipher research, I created a Glossary to ease the way. You may access it here: Research Glossary
Referenced Research Publications
Biological Psychiatry: Global Open Science
2026, marzo 16
DOI: 10.1016/j.bpsgos.2026.100723
The Estrogen–Mitochondria Axis in Bipolar Disorder: From Fluctuating Hormones to Failing Bioenergetics
Abstract
Bipolar disorder (BD) is a psychiatric illness marked by fluctuating mood states and substantial systemic consequences, including dysregulated bioenergetics, immune modulations and significant metabolic dysfunction. Although BD affects males and females at comparable rates, the literature indicates that the symptomatology and course of the disorder can differ between sexes. Females with BD are more likely to exhibit rapid cycling and depressive symptoms, whereas males are frequently associated with reckless behaviour and hallucinations. A plausible mechanism for these symptomatology differences may be marked by the estrogen – mitochondria axis, which reflects the bioenergetic consequences of fluctuating estrogen levels on mitochondrial biogenesis. Estrogen constitutes a family of steroid hormones that have critical regulatory roles in many physiological processes, including reproduction, metabolism, and immune regulation. Within the context of bioenergetics, estrogen supports mitochondrial function in neural tissue by promoting oxidative phosphorylation, reducing oxidative stress, and regulating mitochondrial biogenesis. In this literature review, we examine evidence linking estrogen fluctuation to periods of psychiatric vulnerability across the lifespan in females with BD. There is a particular focus on the mechanistic role of estrogen modulating mitochondrial biogenesis, the existing experimental and pre-clinical evidence underlying this mechanism and the evaluation of current pharmacological and nutraceutical therapeutics that have the potential to modulate this axis. Finally, this review discusses the clinical and future therapeutic implications for behavioural symptomatology across the lifespan of females with bipolar disorder. While this review focuses solely on literature in BD, the plausibility of investigating this mechanism extends to other mood disorders and psychiatric diseases.
Reference
Zachos, K. A., Behravan, K., Demyati, J., & Andreazza, A. C. (2026). The estrogen–mitochondria axis in bipolar disorder: From fluctuating hormones to failing bioenergetics. Biological Psychiatry: Global Open Science. Advance online publication. https://doi.org/10.1016/j.bpsgos.2026.100723
Current Directions in Psychological Science
2025, octubre 22
DOI: 10.1177/09637214251380214
Psychological Science at the Cellular Level: Mitochondria’s Role in Health and Behavior
Abstract
The biopsychosocial model (BPSM) has guided psychological science for decades by emphasizing the interplay of biological, psychological, and social factors in health. Despite its influence, the model has been critiqued for lacking mechanistic specificity, particularly in clarifying how psychosocial factors translate into cellular and molecular processes. Mitochondria, once viewed mainly as energy producers, are now understood to shape immune signaling, stress responses, and neural functioning. Emerging evidence shows that mitochondrial processes are sensitive to psychosocial influences such as chronic stress, trauma, and social connection, with downstream effects on inflammation and disease risk. We highlight findings that support these links while noting areas in which evidence remains preliminary, including possible feedback from mitochondrial changes to psychosocial processes. We also review interventions such as physical activity, mindfulness, and social support that may enhance mitochondrial function. This article positions mitochondria as one promising biological bridge between mind and body, offering a testable extension of the BPSM and identifying opportunities for psychologists to advance this emerging field.
Reference
Fagundes, C. P., Wu-Chung, E. L., & Heijnen, C. J. (2026). Psychological Science at the Cellular Level: Mitochondria’s Role in Health and Behavior. Current Directions in Psychological Science, 35(2), 100-109.https://doi.org/10.1177/09637214251380214
International journal of molecular medicine
2025, octubre 01
DOI: 10.3892/ijmm.2025.5656
Mitochondrial dysfunction in perimenopausal mood disorders: From hormonal shifts to neuroenergetic failure
Abstract
Perimenopause represents a key transition from a reproductive to non-reproductive state in women, characterized by physiological and psychological changes. Mood disturbances during this period, such as depression, anxiety and cognitive decline, are increasingly understood as complex neuroendocrine and metabolic disorders. Mitochondrial homeostasis carries out a key role in the pathophysiology of these affective symptoms. Disruptions in mitochondrial biogenesis, mitophagy and calcium regulation contribute to synaptic dysfunction and neuroimmune changes. These mitochondrial alterations interact with inflammatory pathways and hormonal signals, exacerbating neuropsychiatric symptoms. A more comprehensive understanding of the molecular mechanisms of mitochondrial dysfunction in menopausal mood disorders unveils potential therapeutic strategies, including mitochondria-targeted antioxidants, hormone replacement therapy, and lifestyle interventions designed to restore mitochondrial integrity and cerebral bioenergetic function.
Reference
Yu, Y., Yapeng, H., Liu, Z., Fang, L., Li, J., Luan, Y., Li, W., Cong, H., & Wu, X. (2025). Mitochondrial dysfunction in perimenopausal mood disorders: From hormonal shifts to neuroenergetic failure (Review). International journal of molecular medicine, 56(6), 215. https://doi.org/10.3892/ijmm.2025.5656
Frontiers in neuroendocrinology
2026, enero 19
DOI: 10.1016/j.yfrne.2026.101233
Pathogenesis and potential therapies for perimenopausal depression: Insights from the estrogen-gut microbiota axis
Abstract
Perimenopause represents a critical phase during which women are particularly susceptible to depression. Although fluctuations in estrogen levels resulting from ovarian aging and imbalances in the gut microbiota have been identified as contributing factors to the onset of depression, the interplay among these elements is frequently overlooked. Fluctuations in estrogen levels can further influence neurogenesis or apoptosis through effects on neurotransmitter balance, neuroinflammation, neuroendocrine regulation, and mitochondrial function. Meanwhile, dramatic shifts in estrogen levels can diminish microbial diversity and stability, thereby disrupting the homeostasis of metabolites and neurotransmitters via the gut-brain axis (GBA). Such disturbances may induce neuroinflammation, potentially leading to or exacerbating depressive symptoms. Additionally, the estrobolome (gut bacterial genes encoding estrogen-metabolizing enzymes) plays a regulatory role in the reabsorption, excretion, and systemic levels of estrogen through the modulation of β-glucuronidase activity, thereby affecting estrogen homeostasis. This review first examines the influence of fluctuations in estrogen levels on the composition and function of the gut microbiota, as well as the role of the gut microbiota in estrogen metabolism. It then discusses how estrogen deficiency and dysbiosis of the gut microbiota contribute to the pathogenesis of perimenopausal depression, discussing the potential for a vicious cycle mediated by the estrogen-gut microbiota axis that increases susceptibility to this condition. Finally, this review presents bioactive compounds derived from dietary sources or medicinal plants that exhibit estrogenic and prebiotic properties, which may offer diverse strategies for the prevention and management of perimenopausal depression through modulation of the estrogen-gut microbiota axis.
Reference
Wang, X., Lin, Y., & Zhou, M. (2026). Pathogenesis and potential therapies for perimenopausal depression: Insights from the estrogen-gut microbiota axis. Frontiers in neuroendocrinology, 80, 101233. https://doi.org/10.1016/j.yfrne.2026.101233
Perspectives on Psychological Science
2017, septiembre 22
DOI: 10.1177/1745691617718356
Our (Mother’s) Mitochondria and Our Mind
Abstract
Most of the energy we get to spend is furnished by mitochondria, minuscule living structures sitting inside our cells or dispatched back and forth within them to where they are needed. Mitochondria produce energy by burning down what remains of our meal after we have digested it, but at the cost of constantly corroding themselves and us. Here we review how our mitochondria evolved from invading bacteria and have retained a small amount of independence from us; how we inherit them only from our mother; and how they are heavily implicated in learning, memory, cognition, and virtually every mental or neurological affliction. We discuss why counteracting mitochondrial corrosion with antioxidant supplements is often unwise, and why our mitochondria, and therefore we ourselves, benefit instead from exercise, meditation, sleep, sunshine, and particular eating habits. Finally, we describe how malfunctioning mitochondria force rats to become socially subordinate to others, how such disparity can be evened off by a vitamin, and why these findings are relevant to us.
Reference
Kramer, P., & Bressan, P. (2018). Our (Mother’s) Mitochondria and Our Mind. Perspectives on Psychological Science, 13(1), 88-100. https://doi.org/10.1177/1745691617718356